A) 8960
B) 273
C) 0.120
D) 8.33
E) 1.12 × 10-4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the endothermic reaction A2(g)  2A(g) , a snapshot of an equilibrium mixture of A(g) and A2(g) at low temperature may look as follows. (Each circle represents 1.0 mol of A atoms, and the volume of the box is 1.0 L.)
2A(g) , a snapshot of an equilibrium mixture of A(g) and A2(g) at low temperature may look as follows. (Each circle represents 1.0 mol of A atoms, and the volume of the box is 1.0 L.) 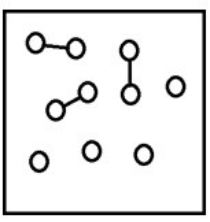 If the system pressure is lowered, what might the new equilibrium system look like?
If the system pressure is lowered, what might the new equilibrium system look like?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At 250ºC, the equilibrium constant, KP, for the reaction PCl5(g)  PCl3(g) + Cl2(g) is 1.80. Sufficient PCl5 is put into a reaction vessel to give an initial pressure of 2.74 atm at 250ºC. What is the partial pressure of PCl5 after the system has reached equilibrium.
PCl3(g) + Cl2(g) is 1.80. Sufficient PCl5 is put into a reaction vessel to give an initial pressure of 2.74 atm at 250ºC. What is the partial pressure of PCl5 after the system has reached equilibrium.
A) 1.50 atm
B) 1.24 atm
C) 4.24 atm
D) 0.94 atm
E) 1.12 atm
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constant, KP, for the reaction H2(g) + I2(g)  2HI(g) is 55.2 at 425°C.
A rigid cylinder at that temperature contains 0.127 atm of hydrogen, 0.134 atm of iodine, and 1.055 atm of hydrogen iodide. Is the system at equilibrium?
2HI(g) is 55.2 at 425°C.
A rigid cylinder at that temperature contains 0.127 atm of hydrogen, 0.134 atm of iodine, and 1.055 atm of hydrogen iodide. Is the system at equilibrium?
A) Yes.
B) No, the forward reaction must proceed to establish equilibrium.
C) No, the reverse reaction must proceed to establish equilibrium.
D) I need to know the volume of the container before deciding.
E) Need to know the starting pressures of all substances before deciding.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
If the system 3H2(g) + N2(g)  2NH3(g) is at equilibrium and more N2 is added, a net reaction that consumes some of the added N2 will occur until a new equilibrium is reached.
2NH3(g) is at equilibrium and more N2 is added, a net reaction that consumes some of the added N2 will occur until a new equilibrium is reached.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction at equilibrium, which one of the changes below would cause the equilibrium to shift to the left? 2NOBr(g)  2NO(g) + Br2(g) , ΔHºrxn = 30 kJ/mol
2NO(g) + Br2(g) , ΔHºrxn = 30 kJ/mol
A) Increase the container volume
B) Remove some NO
C) Remove some Br2
D) Add more NOBr
E) Decrease the temperature
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is correct?
A) If ΔG < 0, then Q > K.
B) If ΔG < 0, then Q < K.
C) If ΔG < 0, then Q = K.
D) If ΔGo < 0, then Q = K.
E) If ΔGo < 0, then Q > K.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is correct?
A) If Q < K, then products must be converted to reactants.
B) If Q > K, then reactants must be converted to products.
C) If Q = K, then the system is at equilibrium.
D) If Q < K, then more reactants are produced.
E) None of the answers is correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction SO2(g) + NO2(g)  SO3(g) + NO(g) , the equilibrium constant Kc is 18.0 at 1200ºC. If 1.0 mole of SO2 and 2.0 moles of NO2 are placed in a 20.0-L container, what concentration of SO3 will be present at equilibrium?
SO3(g) + NO(g) , the equilibrium constant Kc is 18.0 at 1200ºC. If 1.0 mole of SO2 and 2.0 moles of NO2 are placed in a 20.0-L container, what concentration of SO3 will be present at equilibrium?
A) 0.048 mol/L
B) 0.11 mol/L
C) 0.95 mol/L
D) 2.22 mol/L
E) 18 mol/L
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
For ________-________ reactions KP = Kc(RT)Δn gas.
Correct Answer

verified
Correct Answer
verified
Short Answer
When the concentrations of both the reactants and products remain constant because the forward and reverse reactions are occurring at the same rate then the system is ________.
Correct Answer

verified
Correct Answer
verified
Short Answer
Suppose 4.2 mol of oxygen and 4.0 mol of NO are introduced to an evacuated 0.50-L reaction vessel. At a specific temperature, the equilibrium 2NO(g) + O2(g) ![Suppose 4.2 mol of oxygen and 4.0 mol of NO are introduced to an evacuated 0.50-L reaction vessel. At a specific temperature, the equilibrium 2NO(g) + O<sub>2</sub>(g) 2NO<sub>2</sub>(g) is reached when [NO] = 1.6 M. Calculate K<sub>c</sub> for the reaction at this temperature.](https://d2lvgg3v3hfg70.cloudfront.net/TB8482/11eb6c5c_3a93_8765_8d9d_a3579a56b494_TB8482_00.jpg) 2NO2(g) is reached when [NO] = 1.6 M. Calculate Kc for the reaction at this temperature.
2NO2(g) is reached when [NO] = 1.6 M. Calculate Kc for the reaction at this temperature.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constant, Kc, for the reaction PCl3(g) + Cl2(g)  PCl5(g) is 49 at 230°C.
If 0.70 mol of PCl3 is added to 0.70 mol of Cl2 in a 1.00-L reaction vessel at 230°C, what is the concentration of PCl3 when equilibrium has been established?
PCl5(g) is 49 at 230°C.
If 0.70 mol of PCl3 is added to 0.70 mol of Cl2 in a 1.00-L reaction vessel at 230°C, what is the concentration of PCl3 when equilibrium has been established?
A) 0.049 M
B) 0.11 M
C) 0.35 M
D) 0.59 M
E) 0.83 M
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which equation is correct?
A) ΔG = ΔG° - RT logKeq
B) ΔG° = - RT lnK
C) ΔG = RT lnQ
D) ΔG = -RT logQ
E) ΔG° = -RT logKeq
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constants (expressed in atm) for the chemical reaction N2(g) + O2(g)  2NO(g) are KP = 1.1 × 10-3 and 3.6 × 10-3 at 2200 K and 2500 K, respectively.
Which statement is true?
2NO(g) are KP = 1.1 × 10-3 and 3.6 × 10-3 at 2200 K and 2500 K, respectively.
Which statement is true?
A) The reaction is exothermic, ΔHº < 0.
B) The partial pressure of NO(g) is less at 2200 K than at 2500 K.
C) Kp is less than Kc by a factor of (RT) .
D) The total pressure at 2200 K is the same as at 2500 K.
E) Higher total pressure shifts the equilibrium to the left.
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
When a reaction system reaches equilibrium, the forward and reverse reactions stop.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction at 25ºC, is 3 × 1024. 2SO2(g) + O2(g)  2SO3(g)
What is Kc at this temperature? (R = 0.08206 L • atm/K • mol)
2SO3(g)
What is Kc at this temperature? (R = 0.08206 L • atm/K • mol)
A) 1 × 1023
B) 1 × 1024
C) 3 × 1024
D) 6 × 1024
E) 7 × 1025
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction HCONH2(g)  NH3(g) + CO(g) , Kc = 4.84 at 400 K. If ΔH° for this reaction is 29 kJ/mol, find Kc at 500 K.
NH3(g) + CO(g) , Kc = 4.84 at 400 K. If ΔH° for this reaction is 29 kJ/mol, find Kc at 500 K.
A) 5.8
B) 0.17
C) 27.5
D) 0.88
E) 10.3
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct mass-action expression, Qc, for the following chemical reaction? Sn2+(aq) + ½ O2(g) + 3H2O(l)  SnO2(s) + 2H3O+(aq)
SnO2(s) + 2H3O+(aq)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these expressions is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
For any reaction, if ΔG° > 0, then K < 1.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 146
Related Exams